Selected publications
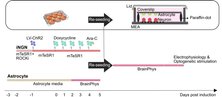
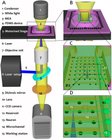
An incubator-independent cell culture system including a double-wall perfusion chamber and ambient-conditioned culture media was engineered and integrated with compartmentalized microfluidic device and multi-electrode arrays (MEAs) electrophysiology devices. Compartmentalized device allowed guidance, tracking and recording from thin axonal branches. Incubator-independent perfusion system enabled continous recording and microscopy. This versatile self-contained system provides long-term and continuous access to the network data .
Habibey R (2023). Incubator-independent perfusion system integrated with microfluidic device for continuous electrophysiology and microscopy readouts. Biofabrication. Accepted. doi: 10.1088/1758-5090/acb466
Morphological & functional dynamics of human neuronal networks on high-density MEAs
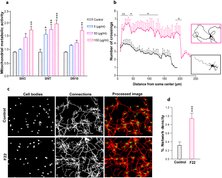
High-density micro-electrode arrays (HD-MEAs) offer non-invasive recording with the best spatial and temporal resolution possible to date. We tracked the morphology and activity features of developing networks derived from human induced pluripotent stem cell (hiPSC) for 3 months. Our morphological data revealed large-scale structural changes from homogeneously distributed neurons in the first month to the formation of neuronal clusters over time. Morphological changes and neuronal movement in the network structure was reflected in corresponding changes of network activity map over time.
Habibey R*, Striebel J*, Schmieder F, Czarske J, Busskamp V (2022). Long-term morphological and functional dynamics of human stem cell-derived neuronal networks on high-density MEAs. Front Neurosci. 16: 951964.
doi: 10.3389/fnins.2022.951964
Microfluidics for neuronal cell and circuit engineering
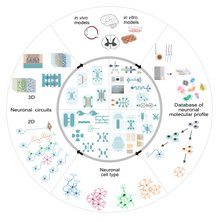
The widespread adoption of microfluidic devices among the neuroscience and neurobiology communities has enabled addressing a broad range of questions at the molecular, cellular, circuit, and system levels. Here, we review biomedical engineering approaches that harness the power of microfluidics for bottom-up generation of neuronal cell types and for the assembly and analysis of neural circuits. Microfluidic 3D neuronal models, together with 2D in vitro systems, are widely used in many applications ranging from drug development and toxicology studies to neurological disease modeling and personalized medicine. Altogether, microfluidics provide researchers with powerful systems that complement and partially replace animal models.
Habibey R*, Rojo Arias JE*, Striebel J, Busskamp V (2022). Microfluidics for Neuronal Cell and Circuit Engineering. Chem Rev. doi: 10.1021/acs.chemrev.2c00212.
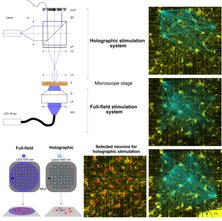
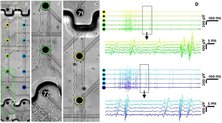
To obtain insights on long-term functional features, a glia–neuron co-culture protocol within MEAs was developed. Full-field optogenetic stimulation enhanced the number of active neurons and their firing rates. Compared with full-field stimulation, which evoked synchronized activity across all neurons, holographic stimulation of individual neurons resulted in local activity. Single-cell holographic stimulation facilitated to trace propagating evoked activities of 400 individually stimulated neurons that revealed precise functional neuronal connectivity motifs.
Schmieder F*, Habibey R*, Striebel J, Büttner L, Czarske J, Busskamp V (2022). Tracking connectivity maps in human stem cell-derived neuronal networks by holographic optogenetics. Life Sci Alliance. 5(7):e202101268. doi: 10.26508/lsa.202101268.
Long-term hiPSCs-derived neuronal networks for electrophysiology and optogenetics
Optogenetic activation of human inducedpluripotent stemcell (hiPSC)-derived neurons facilitates the characterization of their functional properties at the single neuron and circuit level. Here we showcase the preparation of long-term hiPSC-derived neuronal networks expressing optogenetic actuators, in vitro optogenetic stimulation and simultaneous functional recordings using patch clamp and MEA electrophysiology.
Habibey R, Striebel J, Sharma K, Busskamp V (2022). Optogenetic Control of Human Stem Cell-Derived Neuron. Methods Mol Biol. 2501:339-360. doi: 10.1007/978-1-0716-2329-9_17.
Microfluidic device for long-term axonal electrophysiology
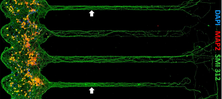
Axons are neuronal compartments critical both for neuronal coding and neurological diseases. Regarding their micrometer size diameter, biophysical measurements of axonal branches are often challenging. Our model allows long-term non-invasive recordings from same axons over months. These data revealed age- and activity-dependent changes in axonal propagation velocity.
Habibey R, Latifi S, Mousavi H, Pesce M, Arab-Tehrany E, Blau A. A multielectrode array microchannel platform reveals both transient and slow changes in axonal conduction velocity. Sci Rep. 2017 Aug 17;7(1):8558. doi: 10.1038/s41598-017-09033-3.
Natural lecithin nanoliposomes promotes neural networks development
Polyunsaturated fatty acids (PUFAs) are critical elements of nervous system structure. Brain function depends on the uptake of “essential” fatty acids such as omega-3 (n-3) and omega-6 (n-6). These PUFAs cannot be readily synthesized by the human body. Here we show that PUFA-rich natural lecithin nanoliposomes increases neurite outgrowth, network complexity, synaptic maturation and neural activity of cortical neurons in vitro. Enhancd neuronal development offeres devising new lecithin delivery strategies for therapeutic applications.
Latifi S, Tamayol A, Habibey R, Sabzevari R, Kahn C, Geny D, Eftekharpour E, Annabi N, Blau A, Linder M, Arab-Tehrany E. Natural lecithin promotes neural network complexity and activity. Sci Rep. 2016;6:25777. doi: 10.1038/srep25777.
Axonal injury and degeneration model on-chip
A miniaturized and thin PDMS microchannel device was combined with a MEA electrophysiology chip and a laser microdissection (LMD) setup to visualize and selectively dissect axonal projections. Microscopy images confirmed progressive antrograde degeneration of distal axonal segments over weeks after focal axonal surgeries. Functional and morphological responses to focal axonal ablations were studied in distinct network compartments at sub-cellular levels and over weeks.
Habibey R, Golabchi A, Latifi S, Difato F, Blau A. A microchannel device tailored to laser axotomy and long-term microelectrode array electrophysiology of functional regeneration. Lab on a Chip, 2015, DOI: 10.1039/C5LC01027F
Engineering 3D modular networks in microfluidic devices
To model 3D modular structure of the brain circuits we coupled thin polydimethylsiloxane (PDMS)-based microchannel devices with multi-electrode array (MEA) chips and assembled 2D and 3D modular cortical networks with predefined structure and available for long-term electrophysiology recordings and microscopy.
Long-term continous electrophysiology from 3D engineered neural networks
A selection of natural gelling agents of non-animal origin were compared for their biophysical properties, neuronal cell embedment and neuronal network survival. Then embeded neuronal networks in guar gum were integrated in our incubator-independent perfusion platform for a proof-of-concept long-term microelectrode array (MEA) electrophysiology study over a period of 79 days.
Wilk N,Habibey R, Golabchi A, Latifi S, Ingebrandt S, Blau A (2016). Selective comparison of gelling agents as neural cell culture matrices for long-term microelectrode array electrophysiology.Oilseeds & Fats, Crops and Lipids, 23 (1).
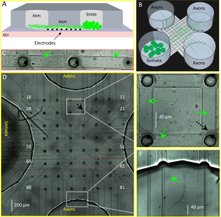
Multicompartment microfluidic device for neural network electrophysiology
PDMS microchannel device on MEAs chips for neural network compartmentalization and co-culture, and axonal guidance and electrophysiology. Cells are loaded in one or multiple reservoirs and axonal branches can take direct or indirect path inside the microchannels. Captured axonal activity in subsequent electrodes is used to measure action potential condunction velocity.
Habibey R, Golabchi A, Blau A. (2015). Microchannel scaffolds for neural signal acquisition and analysis. Springer Series on Computational Neuroscience. 13:47-64.
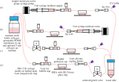
An incubator-independent perfusion platform for automation of the cell culture tasks
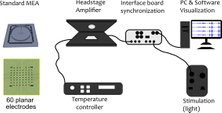
A simple benchtop cell culture perfusion system adapted to commercial microelectrode arrays (MEAs) recording equipment and standard microscope stages for simultaneous and uninterrupted extracellular electrophysiology and time-lapse imaging at ambient CO2 levels. The concept relies on a transparent, replica-molded, gas-permeable polydimethylsiloxane (PDMS) perfusion cap, gravity- or syringe-driven perfusion, and pre-conditioning of pH-buffered serum-free cell culture medium to ambient CO2 levels at physiological temperature.
Saalfrank D, Konduri A, Latifi Sh, Habibey R, Golabchi A, Martiniuc A, Knoll A, Ingebrandt S, and Blau A. (2015). Incubator-independent cell-culture perfusion platform for continuous long-term microelectrode array electrophysiology and time-lapse imaging. R. Soc. open sci. 2 (6), 150031.