Research
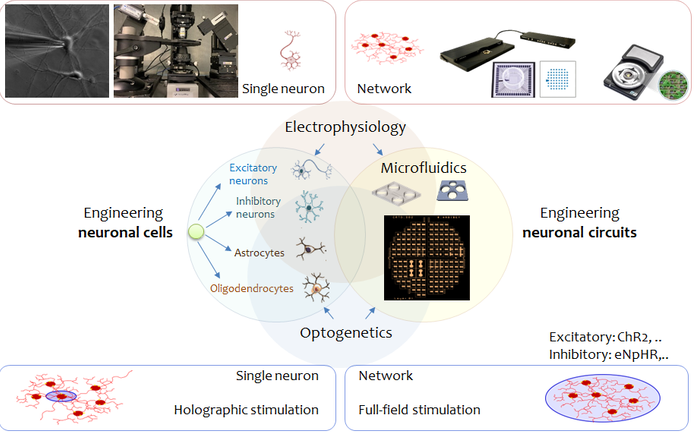
I am fascinated by bottom-up engineering of the in vivo-mimetic neural circuits on-dish/chip. My research lies at the intersection of cellular and network neuroscience, neural tissue engineering, microfluidics and lab-on-a-chip technologies, electrophysiology, and optogenetics. The main goal is to develope functionally robust in vitro platforms for modeling healthy and diseased brain circuits and translate them for biomedical applications like disease modeling, drug development and screening and neural tissue regeneration and replacement therapies.